News
IDT launches improved CRISPR Cas12a enzyme
CORALVILLE, IA (May 30, 2019) — As an advocate for the needs of researchers working at the cutting edge of genomic research, Integrated DNA Technologies (IDT) recently released its latest CRISPR enzyme, Alt-R Cas12a (Cpf1) Ultra.
Cas12a Ultra is a powerful addition to IDT’s Alt-R CRISPR enzyme arsenal—its improved performance makes it easier to achieve precision genome editing at new sites across genomes.
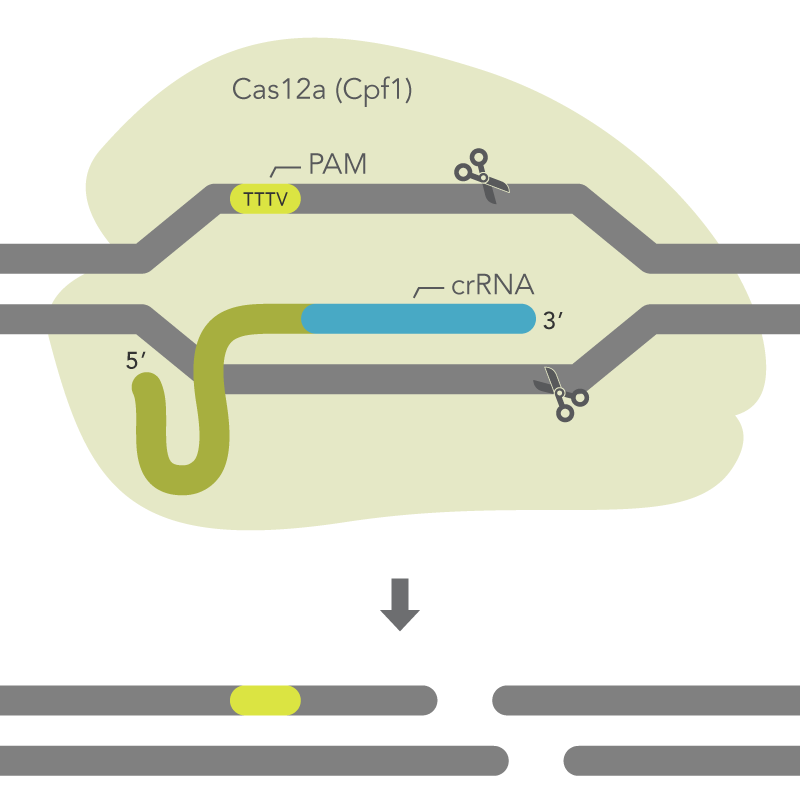
Publication of the Cas12a system in 2015 generated considerable interest among researchers seeking new enzymes for genome editing experiments, particularly due to its unique PAM site (“TTTV”) that greatly expanded the number of potential editing sites in the genome beyond that of Cas9 (“NGG”).
Subsequently, IDT developed a novel mutant of the Acidaminococcus sp. BV3L6 Cas12a (Cpf1), called Cas12a Ultra. The new mutant enzyme has enhanced editing activity, reaching, or exceeding the performance of Cas9. The new Cas12a Ultra also retains activity across a wider temperature range than the wild-type enzyme, making it useful for genome editing in additional organisms, including plants.
IDT’s Alt-R Cas12a (Cpf1) Ultra is a first addition to the genome editing toolbox that offers the same efficiency and versatility as Cas9. The new tool will find equal utility for both knock-out as well as knock-in applications, in fields ranging from basic research to cell engineering to agricultural biotechnology. By incorporating IDT’s Cas12a (Cpf1) Ultra into their research plan, scientists will also benefit from IDT’s quality recombinant genome editing protein manufacturing, high throughput guide RNA synthesis capacity, and best-in-class technical support.
Mark Behlke, IDT’s Chief Scientific Officer, said: “We are proud that IDT continues to take the lead in developing and supplying innovative CRISPR tools to the research community. Alt-R Cas12a (Cpf1) Ultra is a powerful example of this. Researchers will really benefit from the efficiency and versatility that it brings to their gene-editing projects.”
For more information, please visit www.idtdna.com
About IDT
Integrated DNA Technologies, Inc. (IDT) develops, manufactures, and markets nucleic acid products for the life sciences industry in the areas of academic and commercial research, agriculture, medical diagnostics, and pharmaceutical development. IDT has developed proprietary technologies for genomics applications such as next generation sequencing, CRISPR genome editing, synthetic biology, digital PCR, and RNA interference. Through its GMP services, IDT manufactures products used by scientists researching many forms of cancer and most inherited and infectious diseases. IDT is widely recognized as the industry leader in custom nucleic acid manufacture, serving over 130,000 life sciences researchers. IDT was founded in 1987 and has its manufacturing headquarters in Coralville, Iowa, USA, with additional manufacturing sites in San Diego, California, USA; Research Triangle Park, North Carolina, USA; Ann Arbor, Michigan, USA; Leuven, Belgium; and Singapore. For more information, please visit www.idtdna.com and follow on Twitter, LinkedIn, Facebook, YouTube, and Instagram.
Disclaimer: RUO - For research use only. Not for use in diagnostic procedures. Unless otherwise agreed to in writing, IDT does not intend these products to be used in clinical applications and does not warrant their fitness or suitability for any clinical diagnostic use. GMP refers to products manufactured under ISO 13485: 2016 QMS. Purchaser is solely responsible for all decisions regarding the use of these products and any associated regulatory or legal obligations.
Media and PR
For media inquiries please contact IDT Public Relations, idtpr@idtdna.com