Since its beginnings, IDT has worked to improve sustainability practices. Much of the focus has been on manufacturing processes to reduce air emissions, water and energy usage, and hazardous and landfill waste production. Here, we show results from experiments supporting ambient shipping for PrimeTime Gene Expression Master Mix to further mitigate environmental impacts of our products. Eliminating shipping in insulated containers with gel packs and dry ice reduces shipping costs, saving you money. It also lessens detrimental environmental impacts from manufacturing insulated foam containers, addition of dry ice or gel packs, and waste disposal of these items in landfills or incinerators.
PrimeTime Gene Expression Master Mix for probe-based qPCR experiments
For consistency and convenience, many researchers use commercial master mixes that have been developed, often with enhancers and stabilizers, to work in most standard experiments. IDT scientists have developed a flexible qPCR master mix for probe-based experiments for qPCR. The versatile PrimeTime Gene Expression Master Mix produces consistent results under both standard and fast cycling conditions and is compatible with a wide range of real-time PCR instruments.
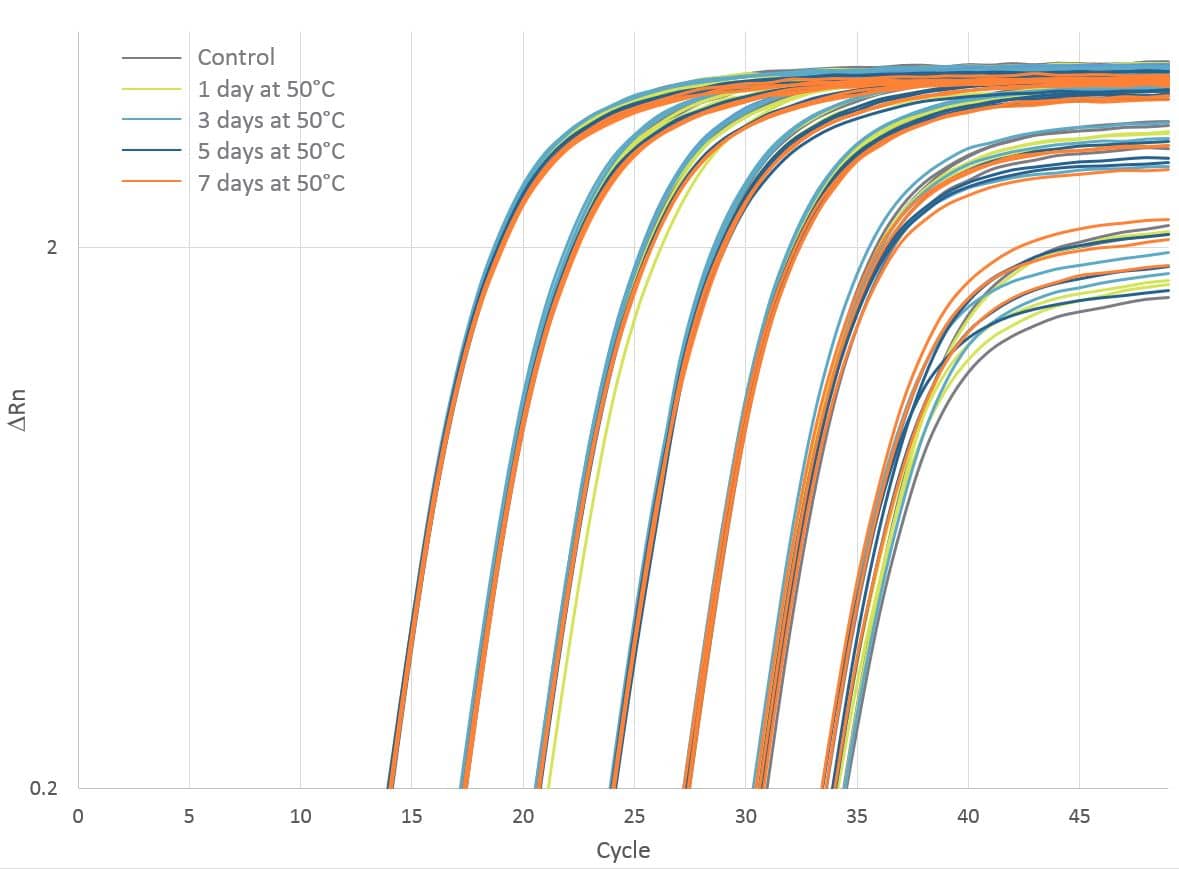
With benchtop stability, PrimeTime Gene Expression Master Mix is compatible with overnight and high throughput experiments. Further extending these thermal stability tests, our research group has shown that master mix function is not affected by conditions that could be encountered during ambient shipping. We continue to observe consistent qPCR results after heat-stressing the master mix at 50°C for up to 7 days (Figure 1) and after 20 freeze-thaw cycles. The master mix retains its ability to provide high-quality results despite exposure to high heat temperature conditions.
Visit www.idtdna.com/qPCRmastermix for more information about the PrimeTime Gene Expression Master Mix. To see additional data from our thermal stability studies, download the white paper, Ambient shipping of PrimeTime Gene Expression Master Mix.
What master mixes do
Table 1 provides a brief description of the role of the main components of a master mix. Note that even small changes in master mix formulation can affect results. Also, some formulations are designed for use with specific cycling conditions (e.g., standard or fast cycling conditions).
| Master mix component | Overview of function |
|---|---|
| Polymerase |
qPCR master mixes normally contain a thermostable DNA polymerase (Taq DNA Polymerase). The polymerase is rendered inactive using either an antibody, chemical modification, or aptamer. The modified polymerases require activation by heating (95°C for 30 sec to 10 min) during the initial denaturation step. PrimeTime Gene Expression Master Mix contains a dual antibody modified Taq polymerase, and is inactive at low temperatures, enabling bench top set up without worry of premature polymerase activity. |
| MgCl2 |
MgCl2 can affect the resolution of the qPCR. It stabilizes primer and probe interactions with DNA and is a cofactor for thermostable DNA polymerases. Typical final MgCl2 concentrations in qPCR can range from 1–6 mM. If MgCl2 levels are too low, polymerase conformation is suboptimal and PCR product yields will be low. If MgCl2 levels are too high, nonspecific amplification and misincorporation of dNTPs occur. |
| dNTPs |
dNTPs are the building blocks for DNA synthesis. Typical reactions contain a final concentration of 10–200 µM of each dNTP. The most effective amount depends on the number of experiments in the reaction (e.g., single-plex or multiplex), the size of the amplicon(s), and the concentration of MgCl2. Lower dNTP concentrations result in increased amplification fidelity but lower yields, compared to higher dNTP concentrations. |
| Reference dye |
Some qPCR instruments require the use of an internal reference dye, such as ROX or a fluorescein dye, for fluorescent signal normalization across wells and to account for pipetting errors. Therefore, certain master mixes are available with different reference dye formulations. Refer to your instrument user manual for instructions. |
| Buffer |
Buffers are required to maintain optimum pH and salt conditions. |
| Additives |
Potential additives include denaturants or proteins that lower Tm or neutralize PCR contaminants from samples to improve resolution or PCR efficiency. |
For research use only. Not for use in diagnostic procedures. Unless otherwise agreed to in writing, IDT does not intend these products to be used in clinical applications and does not warrant their fitness or suitability for any clinical diagnostic use. Purchaser is solely responsible for all decisions regarding the use of these products and any associated regulatory or legal obligations. RUO23-1725_001
























