The experimental design for multiplex quantitative PCR (qPCR) is more complicated than for single reactions. The probes used to identify individual targets must contain unique reporter dyes with distinct spectra. The settings for excitation and emission filters of real-time identification systems vary from manufacturer to manufacturer; therefore, instruments must be calibrated for each dye as part of the experiment optimization process. Ensuring that instruments are appropriately calibrated will enhance dye based qPCR and minimize background and overlap of fluorescent signals.
When selecting dyes:
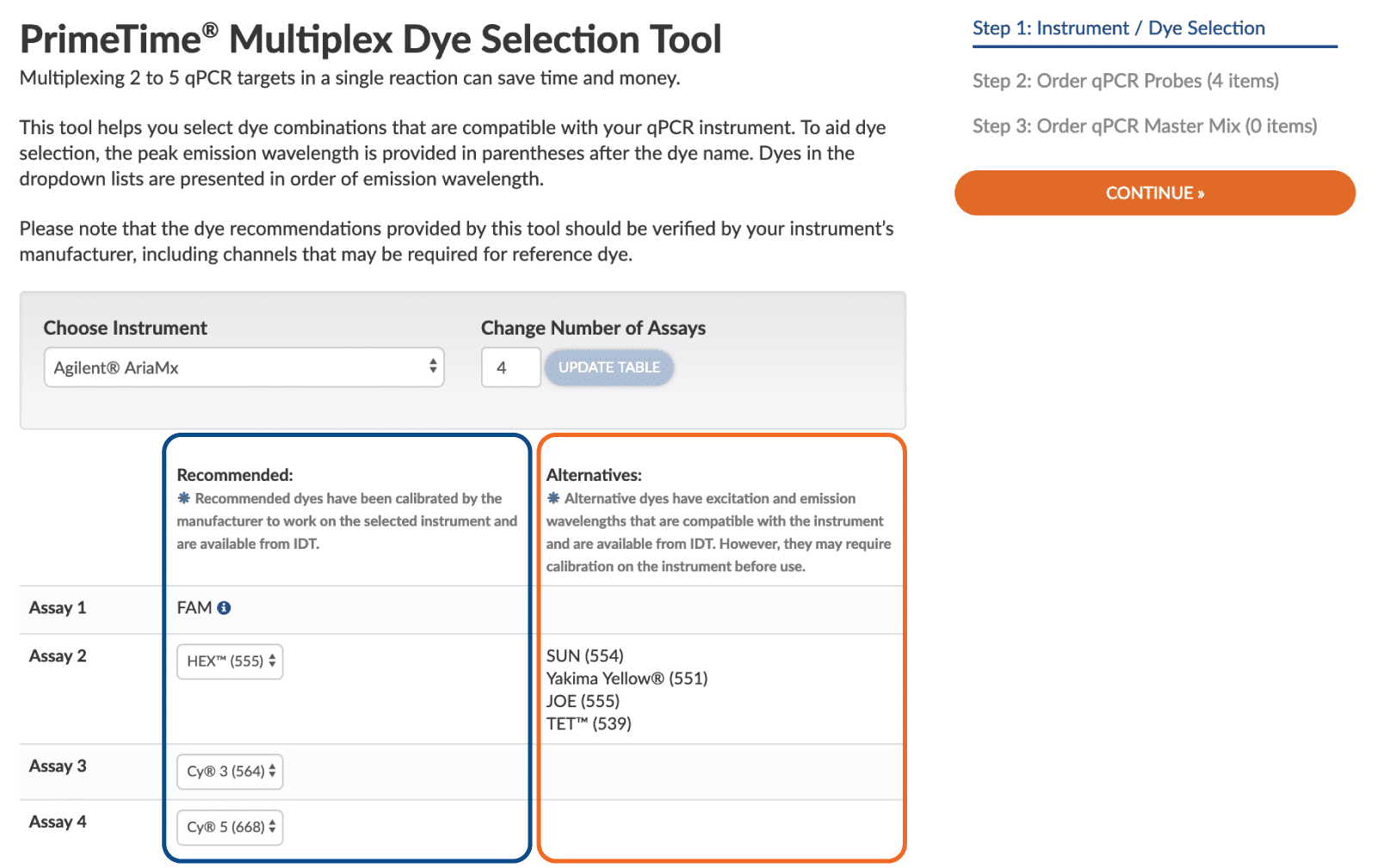
- Choose dyes that are compatible with your instrument. The instrument must be capable of identifying the emission spectrum for each dye you are using. The PrimeTime Multiplex Dye Selection Tool from IDT includes dye compatibility for over 35 different instrument models. After selecting your qPCR instrument from the dropdown menu and selecting the number of assays you are multiplexing, the recommended and alternative dyes are populated. The first column lists the recommended dyes for the instrument (Figure 1, blue boxed content).
- Choose dyes that can be calibrated on your instrument. Your real-time qPCR instrument must be calibrated for the set of dyes you choose. If it is not, you must calibrate the instrument or select different dyes. The second column in the Multiplex Dye Selection Tool offers a list of alternative dyes that are compatible but require instrument calibration (Figure 1, orange boxed content).
- Avoid overlap of emission spectra. Choose dyes with appropriate qPCR dye spectrum with appropriate excitation wavelengths and little to no overlap in their emission spectra. Consider total fluorescence intensity as well. For example, FAM is a good dye choice to identify low-copy transcripts because it has high fluorescent signal intensity. For high-copy transcript (e.g., housekeeping genes), use fluorophores with lower signal intensities. Make sure to avoid any dyes that have overlapping spectra with ROX reference dye, if your instrument uses this as a passive reference dye in qPCR.
Minimize signal cross-talk by using probes with low background fluorescence. Highly efficient dark quenchers, especially those used in combination with a secondary internal ZEN™ or TAO™ internal quencher (Table 1), reduce background fluorescence considerably. Double-quenched probes provide increased reactivity and clear end-point signal, as well as earlier Cq values. This is particularly useful for multiplex reactions, since having several fluorophores in the same tube causes higher background fluorescence. We recommend using the same “quencher type” (all dark quenchers or all fluorescent quenchers) in assays that will be multiplexed.
Table 1. Suggested quenchers for common fluorophores used in multiplex qPCR.
| Fluorescent dye(s) | Excitation wavelength (nm) | Emission wavelength (nm) | Dark quencher(nm) |
|---|---|---|---|
| 6-FAM | 495 | 520 | ZEN/Iowa Black™ FQ |
| SUN™ | 538 | 554 | ZEN/Iowa Black FQ |
| JOE™ | 529 | 555 | ZEN/Iowa Black FQ |
| HEX™ | 538 | 555 | ZEN/Iowa Black FQ |
| MAX™ | 550 | 557 | ZEN/Iowa Black FQ |
| ATTO™ 425 | 439 | 485 | ZEN/Iowa Black FQ |
| Cy® 3 | 550 | 564 | Iowa Black RQ |
| ATTO™ 550 | 554 | 575 | Iowa Black RQ |
| ROX™ | 575 | 608 | Iowa Black RQ |
| Texas Red® -X | 598 | 617 | Iowa Black RQ |
| ATTO™ 590 | 592 | 622 | Iowa Black RQ |
| ATTO™ 647N | 646 | 662 | Iowa Black RQ |
| ATTO™ 700 | 681 | 714 | Iowa Black RQ |
| Cy® 5 | 648 | 668 | TAO/Iowa Black RQ |
| Cy® 5.5 | 683 | 706 | Black Hole Quencher®-3 |
| Yakima Yellow is a registered trademark of ELITech Group. Cy dyes are a registered trademark of Cytiva. Texas Red is licensed from Molecular Probes, Inc (a wholly-owned subsidiary of LifeTechnologies Corporation). JOE is a registered trademark of ThermoFisher. AriaMX is a registered trademark of Agilent Technologies, Inc. Iowa Black, SUN, ZEN, and TAO are trademarks of IDT. | |||
For research use only. Not for use in diagnostic procedures. Unless otherwise agreed to in writing, IDT does not intend these products to be used in clinical applications and does not warrant their fitness or suitability for any clinical diagnostic use. Purchaser is solely responsible for all decisions regarding the use of these products and any associated regulatory or legal obligations. Doc ID: RUO23-1752_001
























