Aldevron CRISPR Nucleases
For clients applying gene editing to investigate and treat genetic and inherited diseases, these nucleases provide an accelerated path to the clinic by offering the same product manufactured at research grade and full cGMP, supported by the quality documentation necessary for regulatory filings.
CRISPR with confidence. We’ll help.
Ordering
Aldevron-manufactured Nucleases
- Available from research through clinical applications
- SpyFi® Cas9 Nuclease displays reduced off-target effects without loss of on-target activity
- Eureca-V™ Nuclease is a type V nuclease based on the Inscripta® novel MAD7® protein that targets a T-rich PAM domain and creates a staggered double-strand break at the target locus (Currently available for research use only)
- SpCas9 Nuclease has proven performance and is manufactured under research use only (RUO) and current good manufacturing practices (cGMP) grades
cGMP Cas Enzymes - Available Now!
Aldevron CRISPR nucleases are designed for seamless transition to the clinic. SpCas9 Nuclease and SpyFi Cas9 Nuclease are available for immediate delivery at cGMP quality. Supported by stability and product quality documentation for regulatory filings.
CONTACT US TODAYProduct details
These nucleases provide a one-vendor strategy for preclinical research through GMP gene editing enzymes, engineered to be used in a variety of applications including electroporation, transfection and microinjection. These products are available at both research and GMP quality grades, providing a seamless transition from research through clinical stage development and commercialization.
SpyFi® Cas9 Nuclease
The SpyFi® Cas9 Nuclease is designed for greater editing efficiency and reduced off-target effects without loss of on-target activity to help accelerate your CRISPR-Cas9 gene editing program. When used in combination with effective guide, target site, and double-stranded DNA cut strategies, SpyFi Cas9 Nuclease can deliver comparable on-target editing and less off-target activity than wild-type SpCas9 nuclease. SpyFi Cas9 Nuclease can satisfy a wide range of needs from basic research to clinical applications . A Drug Master File (DMF) is on file with the US FDA to streamline regulatory filings.
Eureca-V™ Nuclease
Eureca-V™ Nuclease is based on the proprietary MAD7®; nuclease developed by Inscripta®;. Eureca-V is categorized as a Class 2, Type V CRISPR-Cas nuclease. It shares key features common to this family of CRISPR nucleases, including:
- Targets broad array of T-rich PAM domains (YTTV preferred, TTTN functions)
- Requires a single crRNA to cut target DNA sequence
- Staggered double-strand break at cut site creates 5’ overhangs
spCas9 Nuclease
This wild-type spCas9 nuclease is available for use in development work as well as standard GMP products for clinical studies. SpCas9 is ready to use for electroporation, transfections, or injection methods and is supported by an extensive QC panel at research grade and GMP product levels.
Product data
High-efficiency multiplexed T cell editing
Activated human T cells were electroporated with ribonucleoprotein complexes (RNPs) containing SpyFi®; or wild type Cas9 enzymes. High editing efficiency was observed by flowcytometry demonstrating dual knock-out of TCR and PD-1, with comparable editing efficiencies for WT and SpyFi Cas9 RNPs.
Figure 1. High multiplexed knockout efficiency with wild type (WT) and SpyFi RNP. To knockout both TCR and PD-1, activated human T cells were electroporated with an equimolar mixture of RNP targeting TRAC, TRBC, and PD-1. Expression was measured by flow cytometry 5 days post electroporation. Expression was substantially reduced following electroporation with WT or SpyFi RNPs compared to no electroporated cells, n=1.
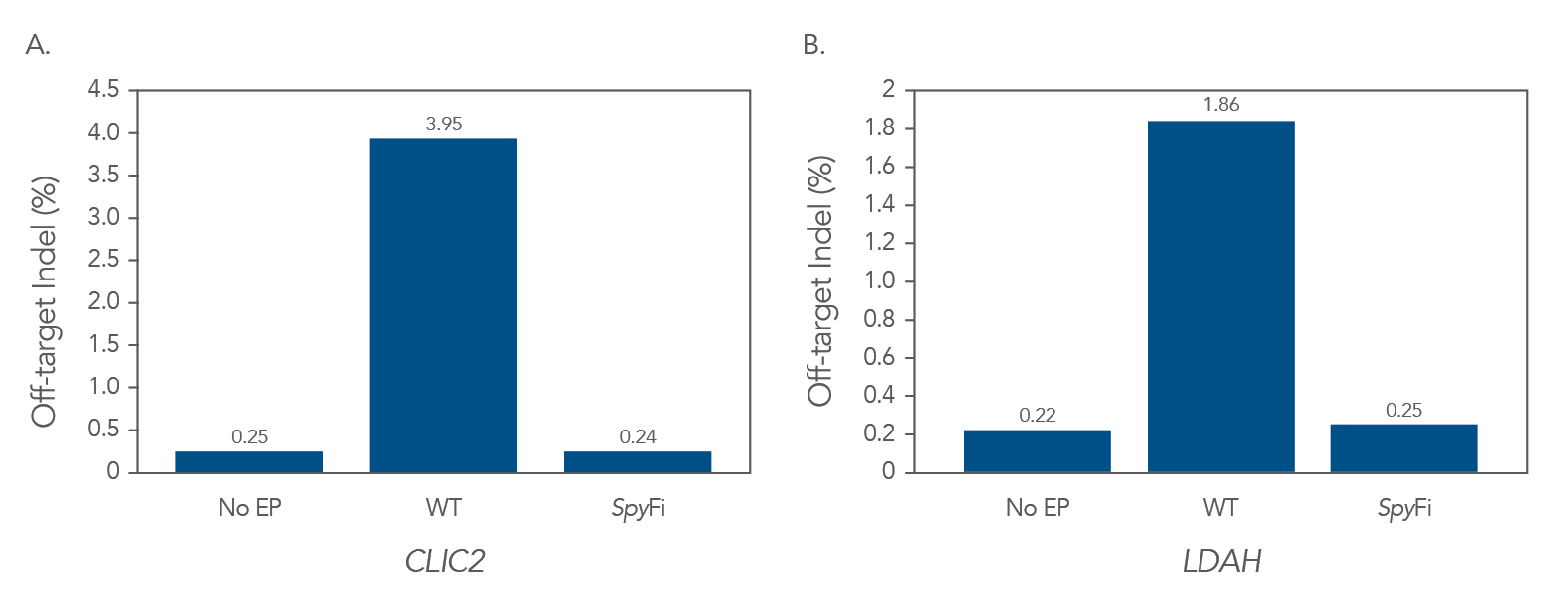
Reduced off-target effects
SpyFi Cas9 reduces off-target effects by up to 94% percent compared to the WT RNP.
Figure 2. Fewer off-target events with SpyFi vs WT RNP. SpCas9 or SpyFi Cas9 RNP targeting the TRAC locus was transfected into activated human T cells using Maxcyte electroporation (EP) technology. The frequency of off-targeting editing for reach RNP was determined by measuring InDel percentages at four off-target binding sites previously identified by iGUIDE [1] . DNA flanking off-target binding sites in CLIC2, LDAH, ANKS1B, and ADCY10 were amplified by PCR, gel-purified, and sequenced by next generation sequencing (NGS). (A) At the CLIC2 locus SpyFi RNP reduced off target editing by 94% compared to WT RNP. (B) Similarly, at the LDAH locus there was an 87% reduction in off-target editing. At both these loci SpyFi had off-target effects comparable to no electroporation controls. At two other sites (ANKS1B and ADCY10), off-target editing was generally less frequent but was diminished by SpyFi (33% less at ANKS1B locus, 15% less at ADCY10, data not shown), n=1.
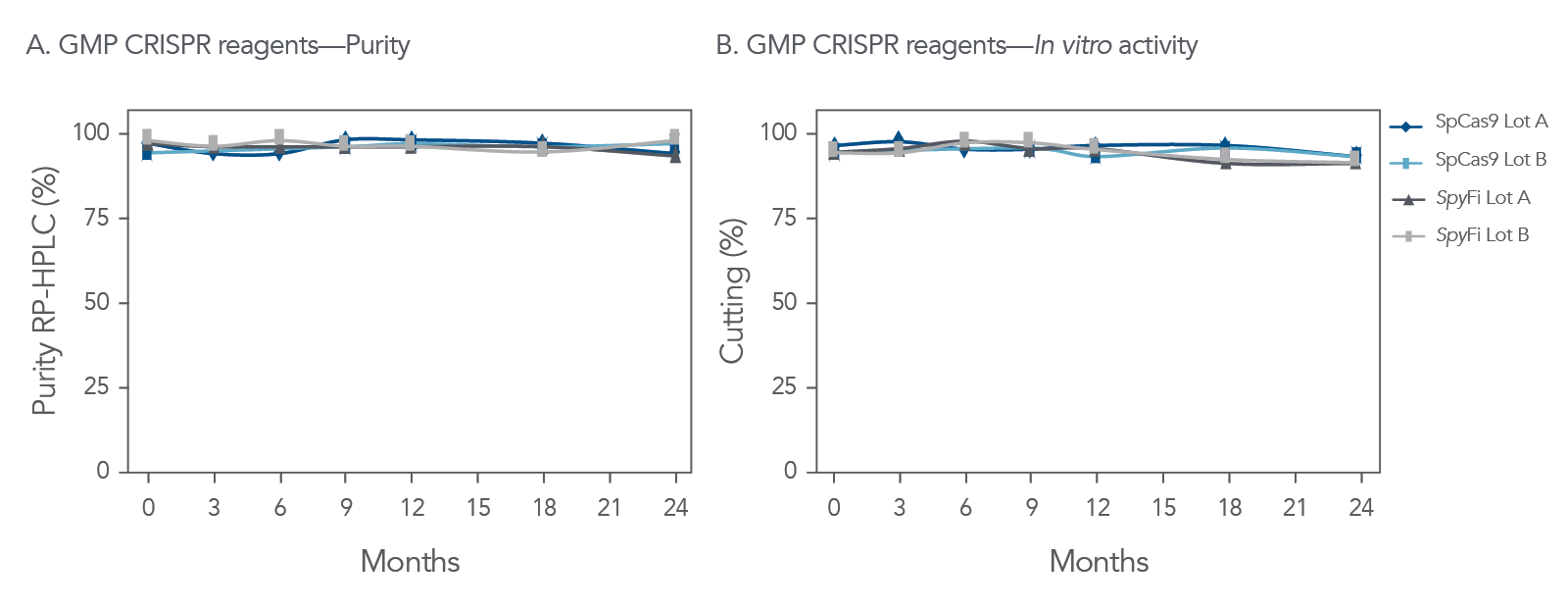
Consistent and reliable manufacturing
Aldevron’s high-quality manufacturing processes provide researchers with consistent and reliable SpCas9 enzymes, helping to enable their next breakthrough. Aldevron’s ISO13485 certification means that research grade Cas9 enzymes can be produced using controlled processes, providing researchers with access to the same, reliable enzymes for their gene editing applications from initial discover to commercialization.
Figure 3. Long-term stability studies demonstrate lot-to-lot consistency and performance over time. (A) Quality tests show no loss of purity or (B) protein activity when the nuclease is stored under recommended conditions. Data depicts two representative lots each of GMP SpCas9 Nuclease and SpyFi Cas9 Nuclease. Aldevron CRISPR Nucleases are stable for 24 months at −20°C. SpCas9 and SpyFi Cas9 Nuclease are formulated at 10.0 mg/mL in 25 mM Tris, 0.3 M NaCl 0.1 mM EDTA, 50% glycerol, pH 7.4, n = 2.
Resources
Frequently asked questions
How would choosing one of these nucleases help my program progress to the clinic?
Are the RUO and GMP products the same?
Where are the products manufactured?
What is the shelf life of the product?
What are the key differences between SpyFi® and Eureca-V™?
What is the lead time for GMP product?
Can Aldevron provide necessary manufacturing information to support regulatory filings?
Can Aldevron provide regulatory expertise to support filings?
Can you share details on the analytical methods used to release the product?
References
- Stadtmauer EA, Fraietta JA, Davis MM, et al. CRISPR-engineered T cells in patients with refractory cancer. Science. 2020;367(6481).
Unless otherwise expressly indicated in a Certificate of Analysis, label or other documentation accompanying the Products, the Products are intended for research use only and subject in each and every case to the condition that such sale does not convey any license, expressly or by implication, to manufacture, duplicate or otherwise copy or reproduce any of the Products.
SpyFi is the property of Aldevron and all information and data presented are solely those of Aldevron. Unless agreed to in writing, IDT does not warrant the fitness or suitability of these products. Purchaser is solely responsible for all decisions regarding the use of these products and any associated legal or regulatory obligations.
-and-spyfi-rnp.png?sfvrsn=1f8af707_6)