Takeaway: Since declared as a global pandemic over 40 years ago, research teams around the world have investigated the human immunodeficiency virus (HIV) to better understand the virus and develop more effective treatments. Now, with the aid of xGen™ NGS amplicon technology, these teams will benefit from more complete coverage, saving critical time and costs.
The human immunodeficiency virus (HIV), is defined by the U.S. Centers for Disease Control and Prevention (CDC) as a virus that attacks the body’s immune system by destroying cells that fight disease and infection. The destruction of the immune system causes acquired immune deficiency syndrome (AIDS). People living with HIV require lifelong medical care to prevent opportunistic diseases that can cause death due to a compromised immune system. To date, the World Health Organization (WHO) reports approximately 40.1 million lives have been lost due to AIDS.
Where did HIV come from and how does it spread?
First identified in chimpanzees in Central Africa, HIV’s zoonotic origin is believed to have started with the transfer of the Simian immunodeficiency virus (SIV) from primates to humans, most likely through exposure to infected ape blood and/or bodily fluids. Over the subsequent decades, HIV spread throughout African villages and eventually in other parts of the world. For the last 40 years, the virus has been classified as a global pandemic.
Unlike SARS-CoV-2, transmission does not occur through respiratory particles, but through the exchange of bodily fluids (i.e., blood, semen, breastmilk) from an infected individual to an uninfected individual, similar to what health officials are seeing with the monkeypox virus. A person can be infected but asymptomatic, which increases the risk of transmission.
Stages of HIV
Once an individual has contracted the virus, there are three stages of the illness:
Stage 1: Acute HIV infection
The individual will experience flu-like symptoms―fever, sore throat, muscle aches, fatigue, rash, etc.―due to a large amount of HIV in their blood and is considered highly contagious.
Stage 2: Chronic asymptomatic HIV infection
As the illness progresses, HIV remains active throughout the body, but symptoms may reduce, with some individuals not experiencing any symptoms. While the person may not feel ill, they are still highly contagious and will continue to spread the virus if engaged in high-risk activities.
Without medical intervention, this stage may go on for a decade or more, or could progress faster for some. When nearing the end of this phase, the HIV load within the blood will increase, moving the individual to the third and final phase.
Stage 3: Acquired immunodeficiency syndrome (AIDS)
Health officials estimate the history of HIV in the United States began in the early 1970s, but it wasn’t until June of 1981 that the first reported case of AIDS was recorded.
Once diagnosed with AIDS, the patient experiences a high viral load, may easily transmit HIV, and will experience an increasing number of infections or other serious illnesses. Without treatment, the CDC reports most individuals survive for only three years.
HIV research: Past and present, and future
While considerable progress has been made since the first known case of HIV in humans, HIV infection remains a persistent public health concern across the globe. According to annual WHO statistics, there were an estimated 38.4 million people living with HIV worldwide at the end of 2021, 1.5 million new cases were diagnosed, and 650,000 individuals died from the illness.
Advancements in treatment offerings have enabled patients to manage HIV, reduced the number of cases that progress to AIDS, and prolonged average life expectancy. However, without an effective cure, people living with HIV continue to suffer from this disease and require medical care throughout their lives, leaving scientists looking for new prevention and therapeutic options.
xGen amplicon technology for HIV research
The HIV life cycle requires the conversion of the viral RNA genome to DNA using an error prone viral reverse transcriptase (RT). Because the HIV RT is low-fidelity, in vivo replication introduces 5-10 errors per genome. The rapid mutation rate of HIV is a major factor in the ability of the virus to develop drug resistance. It also presents challenges for targeted sequencing using amplicons because mutations arising at primer sites will cause primer drop-out and subsequent read loss.
The COVID-19 pandemic demonstrated that IDT’s xGen NGS amplicon technology was capable of sequencing SARS-CoV-2 variants, and the xGen SARS-CoV-2 Amplicon Panel was resilient to coverage drop out due to viral mutations. NGS research and development teams at IDT set out to determine if the technology used for the xGen SARS-CoV-2 Amplicon Panel would also provide NGS sequencing solutions for a virus with an even higher rate of mutation: HIV.
The team, led by Laurie Kurihara, Senior Director of Product Development, designed a HIV amplicon panel against the reference genome HXB2 (GenBank: K03455.1) using 109 overlapping amplicons and tested it against four HIV variants with differing identities to the design genome. Initial testing of the panel was conducted with HIV variant 1, which has 93% identity with HXB2. Variant 1 had genomic coverage of 96%, with high on-target rates of >95% in the presence of 10 ng human gDNA.

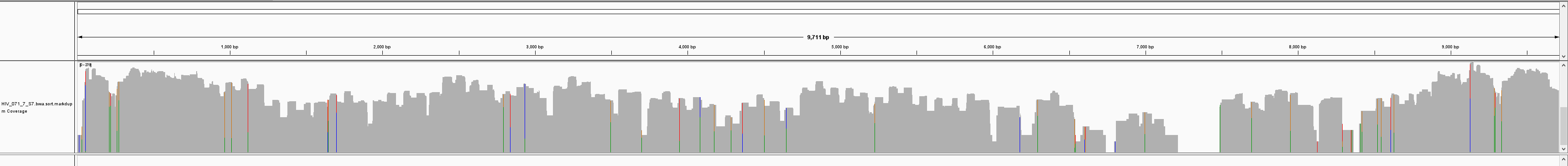
By sequencing longer reads (PE250), the team saw improved coverage due to super amplicons (of 2x and greater) that are generated by the unique xGen amplicon technology.
PE150:
PE250:
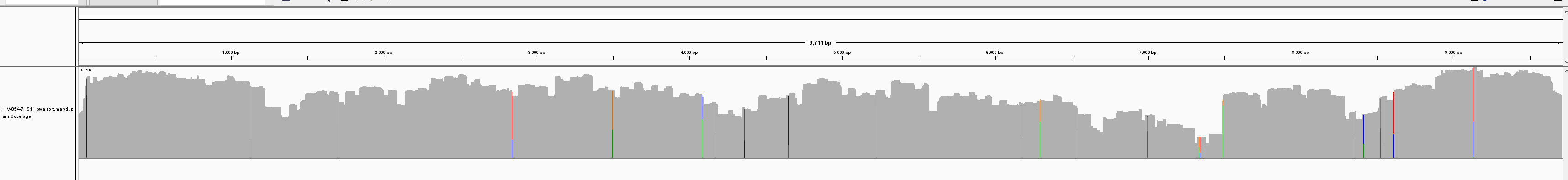
Table 1:
| % Coverage | |||
|---|---|---|---|
| Variant | % Identity (HXB2) | PE 150 | PE 250 |
| 1 | 93 | 96 | 100 |
| 2 | 92 | 93 | 98.8 |
| 3 | 91 | 88 | 100 |
| 4 | 93 | 95 | 99.6 |
Subsequent experiments comparing four HIV variants showed improved coverage with PE250 (Table 1). Overall, sequencing with PE250 resulted in greater than 98% genomic coverage for four HIV variants with varying degrees of sequence identity. This demonstrates the ability of xGen amplicon technology to generate full coverage for challenging genomes with high mutation rates, such as HIV—offering researchers a powerful new tool for HIV research.
xGen NGS―made for infectious disease research.
*RUO—For research use only. Not for use in diagnostic procedures. Unless otherwise agreed to in writing, IDT does not intend for these products to be used in clinical applications and does not warrant their fitness or suitability for any clinical diagnostic use. Purchaser is solely responsible for all decisions regarding the use of these products and any associated regulatory or legal obligations.